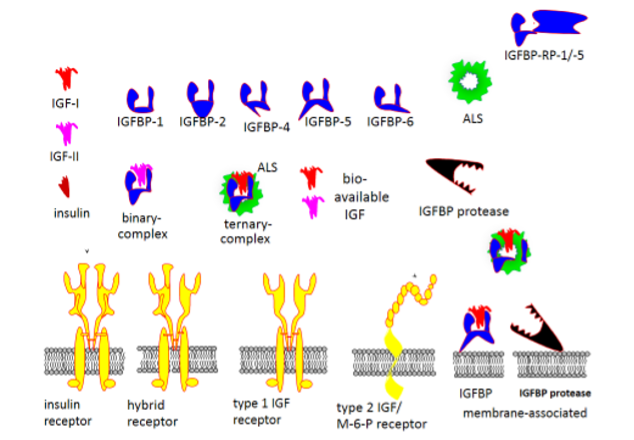
Figure 1. The primary structures of IGF-I, IGF-II and insulin are similar. IGFs are produced by many differentiated cell types, and their bioactivity in the extracellular fluids or in the circulation are coordinated by six IGF binding proteins (IGFBP-1 through -6). IGFBP-3, the major binding protein in serum is stimulated by GH and it forms a large 150 kDa ternary complex with IGF-I or -II and the GH regulated acid labile subunit (ALS). IGFBP-5, an important supporter of bone tissue formation, also forms ternary complexes with IGF-I or -II and ALS. IGFBP-1, suppressed by insulin, is one of several binding proteins in the smaller 50 kDa binary complexes with IGF-I or –II. IGFBP-2 has inverse association with insulin under many physiological conditions. In contrast, IGFBP-4, -5 and -6 do not appear to be directly regulated by GH or insulin and are important local regulators of IGF activity in bone and the CNS. The type 1 IGF receptor (IGF1R) is the mediator of the mitogenic, anti-apoptotic, differentiating and metabolic effects of both IGF-I and -II. The structural similarity of the IGF1R with the insulin receptor (IR) explains the formation of hybrid receptors in cells that expresses both receptors such as myocytes and pre-adipocytes. Cross reactivity among the ligands and the receptors have been demonstrated, although it has minor importance under physiological conditions but may cause non-islet-cell tumor hypoglycemia due to unprocessed pro-IGFs with markedly decreased binding affinity to IGFBPs. A second receptor, exclusively binding IGF-II, work as a scavenger receptor and is identical to the mannose-6-phosphate receptor, known to direct proteins for degradation in the lysosomes. A second level of control of IGF bioactivity is exerted by IGFBP proteases which release IGF-I activity after fragmentation of IGFBPs. Specific production and regulation of IGFBP proteases at the tissue level controls processes such as ovulation and atherosclerosis. Furthermore, interaction of IGFBPs and IGFBP proteases with the extracellular matrix modify the binding affinity for the IGFs and are involved in prolonging the actions of IGFs at the tissue level. Extracellular matrix also signals though integrin receptors on the cell surface and modifies IGF-1R signaling. This figure also shows the existence of IGFBP-related proteins with markedly lower affinity for the IGFs and with physiological roles not related to their IGF binding.
